- Means of data acquisition (uploaded December 2014);
1.a. Active versus passive surveillance
1.b. Enhanced passive surveillance
1.c. Syndromic surveillance - Surveillance objective (uploaded July 2015);
2.a. Surveillance purpose versus objective
2.b. How to identify the appropriate surveillance objective?
2.c. Specific characteristics of individual surveillance objectives - Evaluation (uploaded August 2015);
- Risk-based surveillance (to be uploaded).
Working group: It was emphasized that the classification as active or passive is done on the basis of how the data is obtained, not what action1 follows. Therefore, it was agreed that it is acceptable to talk about passive surveillance, which is also common practice (Garcia-Abreu et al. 2002, WHO 2005, Dufour and Hendrikx 2009, Cameron 2012, Hoinville 2013).
1Action in this sense does not only refer to control measures, but also to whether further tests are preformed.
The emphasis of passive surveillance lies on the observer-initiated provision of animal health related data (e.g. voluntary notification of suspect cases). Hence, the sensitivity of passive surveillance depends on many factors, including:
- probability of infected animals showing detectable clinical signs;
- the disease awareness of farmers, veterinarians and health authorities;
- their motivation to report; and
- the sensitivity of any diagnostic procedure used.
Passive surveillance relies on the detection of clinical signs and is thus not designed to detect subclinical disease. The reporting threshold to contact a veterinarian or authorities (or similar step by which the reporting chain is initiated) is a major limitation to passive surveillance. Therefore, for instance for notifiable diseases, the trust and cooperation between the farmer and the veterinarian, the farmer and the government and the veterinarian and the government affect the sensitivity of passive surveillance. In turn, measures to strengthen trust can improve the sensitivity, such as prompt payments of compensation or keeping negative effects of precautionary measures (e.g. movement control, culling) for the farmer to a minimum until disease suspicion is confirmed.
Active surveillance, in contrast, is initiated by the investigator, fulfilling the following criteria:
- Process that results in animals or herds being subject to surveillance is initiated by and under control of the investigator,
- Surveillance activities are scheduled in advance (pre-determined),
- Planned selection of animals or data based on a defined sampling frame (regardless if sample or census).
-
What action follows (e.g. whether a sample is collected after a case has been identified through a passive component),
-
Whether reporting is mandatory (notifiable diseases) or not,
-
Whether only a sub-set or all animals (census) are sampled, or
-
What the likelihood of detecting an infected animals is.
Typical examples of active and passive surveillance are:
| Active surveillance | Passive surveillance |
|---|---|
|
Observer (e.g. farmer, veterinarian, hunter, general public) notices clinical signs, which is subsequently reported to the investigator (by original observer or intermediate(s)). |
Potentially tricky examples were selected to illustrate the distinction between active and passive surveillance:
| No. | Hazard | Description of component | Classification | Justification |
| 1 | Classical swine fever |
Sampling of hunted, healthy wild boar (see 2002/106/EC: Chapter IV, section H(3)) |
Active |
|
| 2 |
Sampling of wild boar shot sick or found dead (by hunter) (see 2001/89/EC: Article 15(2)(c)) |
Passive |
|
|
| 3 | Sampling of wild boar involved in accidents | Passive |
|
|
| 4 | Tuberculosis |
Examination of carcasses at the abattoir for TB lesions (see 64/432/EWG; Annex A, Section I(2c)) |
Active |
|
| 5 | Avian influenza |
Testing of healthy wild birds captured or hunted (see SANCO/10268/2006, Annex II, section B) |
Active |
|
| 6 | Scrapie |
Testing of slaughtered sheep (abattoir) (see Regulation 999/2001, Annex II (2)) |
Active | See No. 4 |
| 7 |
Testing of deceased sheep (rendering plant) (see Regulation 999/2001, Ch. A, Annex II(3)) |
Active |
|
|
| 8 |
Testing of sheep with neurological signs (farm; rendering plant) (see Regulation 999/2001, Article 3(1h)) |
Passive | See No. 2 | |
| 9 | Bluetongue | Clinical examinations of 150 herds per geographical unit; sample collection in case of clinical signs | Active |
|
“Active investigator involvement” in this context refers to any actions by the investigator to increase the likelihood of reporting (“observer-initiated provision of animal health data”) either due to improved recognition of clinical signs or increased preparedness to report.
It was agreed that the following criteria may act as enhancements to passive surveillance (justification):
-
Payment of rewards for notifications (financial incentive to report)
-
Training (improves ability to recognize clinical signs and awareness to report)
No full consent could be reached for the following discussed criteria (justification; number of working group respondents who agreed/disagreed – “don’t know” responses were not counted):
-
Awareness campaigns (improves ability to recognize clinical signs and awareness to report; 6/2)
-
Payment of compensation for the losses the farmer incurs after a notifiable disease has been confirmed (financial incentive: 6/2)
-
Provision of alternative routes of reporting such as a phone hotline or notification by SMS (simplified reporting; 6/1)
-
Payment of the testing cost or some other form of mutual agreement such as farmer receiving advice in return (incentives to report; 4/2)
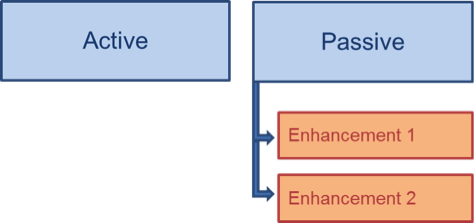
Since criteria may not be clear-cut and more than one enhancement can apply, it may be appropriate to specify which enhancements were applied and identify which step in the reporting process the enhancement aims to influence (Figure 1a and Figure 2), rather than distinguishing between passive and enhanced passive components (Figure 1b).
 |
 |
Figure 1. Grouping level of enhanced passive surveillance in relation to active and passive surveillance: a) Vertically = distinction between active and passive surveillance only, with specification of enhancements under passive surveillance (if applicable); b) Horizontally = distinction between active, passive and enhanced passive surveillance.
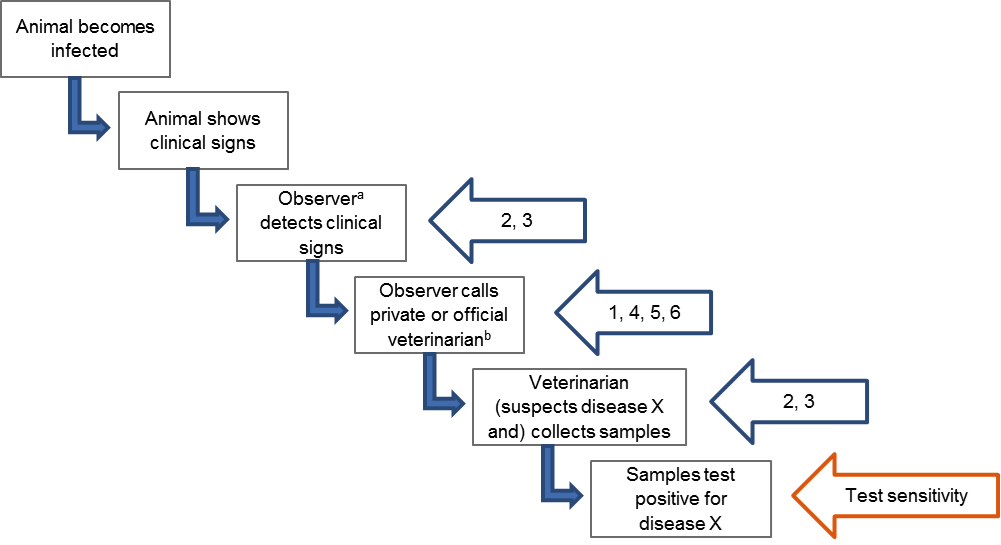
Figure 2. Schematic diagram illustrating steps leading to reporting of Disease X based on passive clinical surveillance and which of these steps are targeted by the six enhancements listed under FAQ4: 1. Rewards for notifications, 2. training, 3. awareness campaigns, 4. compensation for losses the farmer incurs, 5. alternative routes of reporting, 6. payment of testing cost or other kind of mutual agreement. Beside these enhancements, test sensitivity is an additional factor influencing sensitivity of passive clinical surveillance.
a Observer can be a farmer, hunter, member of the general public, private veterinarian, ….
b Private veterinarian can be the investigator (e.g. if responsible for herd-level surveillance of Disease X) or an intermediate who will notify the investigator (e.g. by submitting samples to an official laboratory or notifying authorities).
Legal requirements to increase the likelihood of reporting may exist in various forms, e.g.
- Mandatory reporting for notifiable diseases;
- Legal requirements to collect a minimum number of samples through passive surveillance;
- Obligation to carry out diagnosis of exclusion if certain criteria apply: e.g. exclusion of brucellosis if abortion occurs during last third of pregnancy in cows older than 24 months.
Although these measures are intended to increase the likelihood of reporting, the opposite may actually happen depending on the consequences of reporting. Therefore, it is important to strengthen compliance to legal reporting requirements by carefully assessing measures to improve trust between the observer and the investigator (see FAQ 2).
Further sources of reading
Hadorn and Stärk (2008); Schatz et al. (2014); Enhanced Passive Surveillance project
[Response:] Surveillance of syndromes is related to the type of case definition that is used and not to the modality of reporting. Even though data sources used for syndromic surveillance may appear to be actively (e.g. bi-weekly bulk-milk collections) or passively collected (e.g. clinical data from practitioners, direct lab submission), a key feature of syndromic surveillance is that these data sources are used pre-diagnostically and analysed to identify deviations from a baseline.
Further sources of reading
Dorea et al. (2011), Dupuy et al. (2013), Vial and Reist (2014), TripleS project.
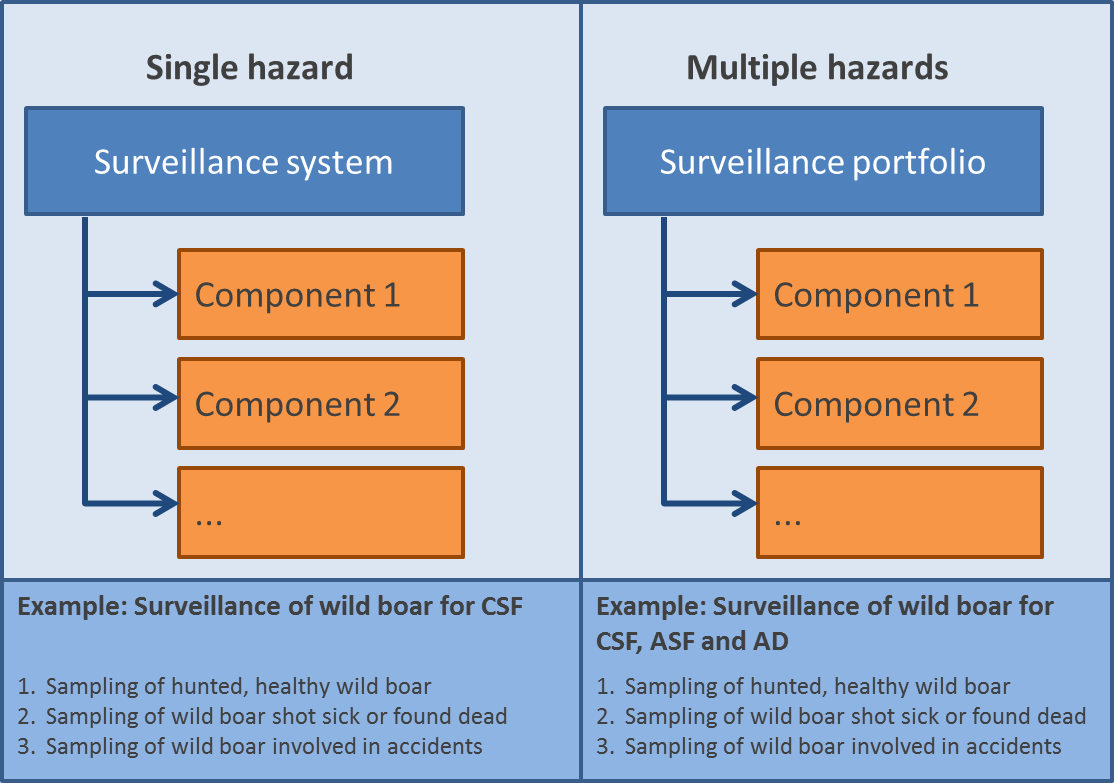 A surveillance component is a single surveillance activity used to investigate the occurrence of one or more hazards (see CSF example at the bottom of Figure 1). Several components make up a surveillance system or a surveillance portfolio depending on whether the occurrence of one or more hazards1 is investigated.
A surveillance component is a single surveillance activity used to investigate the occurrence of one or more hazards (see CSF example at the bottom of Figure 1). Several components make up a surveillance system or a surveillance portfolio depending on whether the occurrence of one or more hazards1 is investigated.
Figure 1 (on the right) shows the distinction between a surveillance system (targeting a single hazard) and a surveillance portfolio (targeting multiple hazards) and illustrative examples. CSF = Classical Swine Fever; ASF = African Swine Fever; AD = Aujeszky Disease.
1Hazard: A biological, chemical or physical agent in, or a condition of, an animal or animal product with the potential to cause an adverse health effect, i.e. any health hazard (e.g. pathogen, toxin) or its risk factors. OIE (2010). Handbook on import risk analysis for animals and animal products. Volume 1: Introduction and qualitative risk analysis. Paris, France, OIE.
Surveillance purpose has a more global meaning compared to surveillance objective. Hence, surveillance purpose should depend on the outputs needed to support decision-making and thus be policy-driven. It should describe why surveillance is necessary and how, when combined with intervention measures, it will impact on public health, animal health or the economy. In contrast, surveillance objective refers to the specific goal(s) to be met when collecting the data (see examples provided under FAQ 3 and FAQ 4).

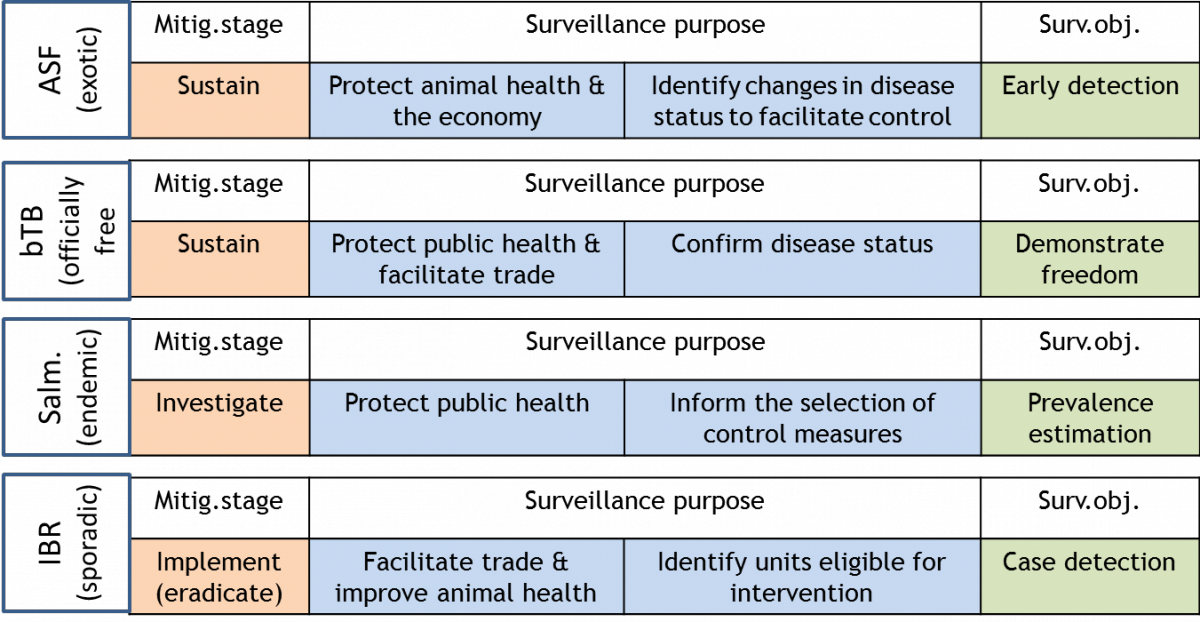
The working group agreed that categories should be available to describe both parts of the surveillance purpose definition. They (partly) agreed on
- eight categories for “what surveillance will accomplish” (Table 2) and
- five categories for “why it is necessary” (Table 3).
It was agreed that surveillance alone cannot achieve the options of “why surveillance is necessary” without the implementation of intervention measures. Therefore, the working group recommends considering (see Figure 2):
- the mitigation stage (sustainment, investigation or implementation) (Häsler et al. 2011)
- both the categories when describing surveillance purpose, i.e.
- “why surveillance is necessary” and
- “what it will accomplish”, and
- the associated surveillance objective (see FAQ4).
|
No. |
Surveillance purpose |
Mitigation stage |
Nagreed / Nvalid |
|---|---|---|---|
|
1 |
To confirm disease status |
Implementation or sustainment |
4 / 6 |
|
2 |
To identify changes in disease status to facilitate early response |
Sustainment |
4 / 6 |
|
3 |
To inform the selection of intervention measures |
Investigation |
5 / 6 |
|
4 |
To inform the planning prioritisation and conduct of research |
Investigation |
5 / 6 |
|
5 |
To inform priorities for disease surveillance and intervention |
Investigation |
5 / 5 |
|
6 |
To provide information for assessing and managing risks |
Investigation |
6 / 6 |
|
7 |
To identify units eligible for intervention |
Implementation |
5 / 6 |
|
8 |
To assess if intervention measures are efficient (monitor progress, verify success) |
Implementation |
4 / 5 |
|
No. |
Reduce the occurrence of disease in animal populations in order to |
Nagreed / Nvalid |
|---|---|---|
|
1 |
Protect the economy or facilitate trade |
5 / 6 |
|
2 |
Protect animal health |
6 / 6 |
|
3 |
Improve animal productivity |
5 / 5 |
|
4 |
Protect public health |
6 / 6 |
|
5 |
Protect animal welfare |
3 / 6 |
The working group agreed to distinguish between four main surveillance objectives that are
- Early detection,
- Freedom from disease,
- Prevalence estimation and
- Case detection
These objectives present goals that when met will result in the collection and analysis of data in order to achieve the purpose of the system. Please note that examples of surveillance objectives provided by the OIE in the Guide to Terrestrial Animal Health Surveillance, page 5 (OIE 2014a) and in the Terrestrial Animal Health Code (section 2.2) (OIE 2014b) appear to be a mixture of purpose and objective as judged by the working group. Further comments from the surveillance community are welcome.
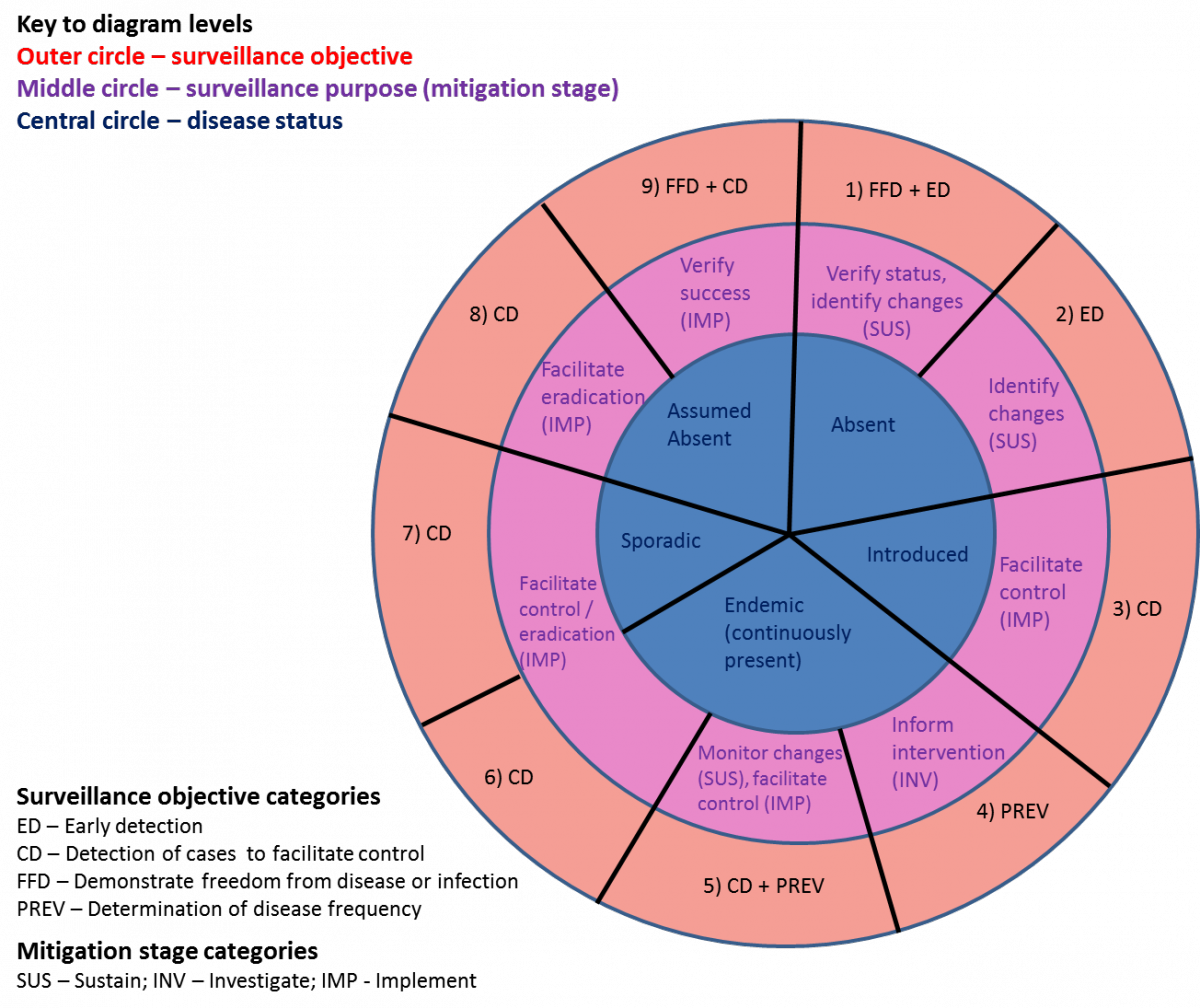
The current hazard situation and surveillance purpose determine potential stages where the surveillance program might be at (Figure 3).
However, not all stages need to be covered in sequence. For instance, successful outbreak control can lead from 3 to 8 as disease is eliminated without becoming endemic. This diagram shall only serve as a guide, but will not reflect every scenario in practice (e.g. not all possible combinations of components).
The following bullet points illustrate the time when the shifts between different stages generally occur:
- From 1 to 2: Disease has been historically absent or absent for a long time. Therefore, residual infection can be ruled out and (theoretically) freedom can be maintained by early detection measures only.
- From 2 to 3:; A known or unknown disease has been introduced. The aim is to control the outbreak.
- From 3 to 4: The outbreak could not be controlled and the disease has become endemic. This stage aims to monitor disease trends and obtain epidemiological information (e.g. risk factors) to inform the assessment and selection of appropriate intervention strategies;
- From 4 to 5: The focus shifts from monitoring changes in disease trends to activities with the aim to control the disease if a change in disease trend occurs;
- From 5 to 6: The focus lies on controlling or eliminating an endemic disease;
- From 6 to 7: The focus lies on controlling or eliminating a disease which has become sporadic due to previous intervention efforts; a decision needs to be made whether to control disease to remain at low levels (stay at 7) or to eliminate it (move to 8);
- From 7 to 8: Final stage of elimination with the aim to detect and eliminate last pockets of infection;
- From 8 to 9: The aim is to verify the success of the elimination efforts;
- From 9 to 1: Elimination has been demonstrated (freedom has been achieved). The aim is now to sustain free status and detect new introductions early.
The working group agreed that ED may apply to hazards present at low levels.
Difference to CD: Case detection aims to detect (individual) cases within an infected population, whilst ED aims to detect new infection in case a population becomes infected (see FAQ 8). Hence, CD focusses predominantly on intervention (mitigation stage: implementation), whilst ED focusses on prevention (mitigation stage: sustainment). Furthermore, CD targets individual animals or herds, whilst ED targets an entire population.
Difference to PREV: Surveillance to detect changes in prevalence (PREV) usually relies on conducting representative surveys. When disease is present at low levels, risk-based approaches may be more appropriate to increase the likelihood of detecting new infections in high-risk strata as quickly as possible in order to ensure rapid implementation of intervention measures with the aim to prevent an extended epidemic and thus minimize negative consequences.
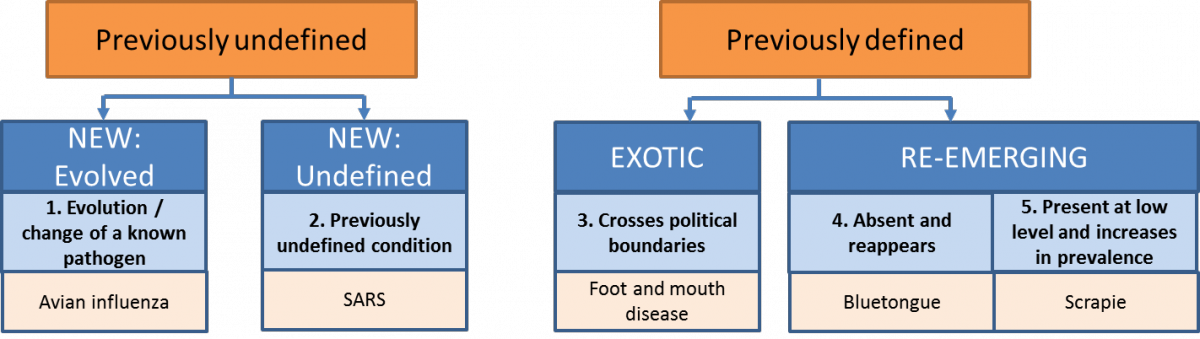
Definition:
Early detection is defined as “surveillance … to increase the likelihood of timely detection of undefined (new) or unexpected (exotic or re-emerging) threats” (Hoinville et al. 2013, Rodriguez-Pietro et al. 2014). According to the definitions of ‘new’, ‘exotic’ and ‘re-emerging’ diseases, ED also includes identifying an increase in prevalence of hazards that are present at low level (no. 5 in Figure 4).
When designing surveillance it is helpful to have one primary surveillance objective in mind to ensure that the surveillance approaches selected are most appropriate for this primary objective. However, a surveillance system with the objective to demonstrate disease freedom generally includes surveillance components with the primary objectives of a) FFD and b) ED in parallel as the ED components can contribute to demonstrating freedom (Figure 5).


Therefore, two aspects have to be distinguished when the objective is to demonstrate disease freedom:
- The likelihood that the hazard is still present in the population = risk of (residual) infection and
- The likelihood that the hazard will be re-introduced = risk of (new) introduction
FFD and ED should always be designed in parallel as they complement each other:
- FFD is designed to demonstrate that a population is not infected and thus to detect (residual) infection whilst ED is designed to detect new introductions;
- FFD usually relies on ad hoc surveys, whilst ED often involves continuous surveillance or surveys in short intervals; hence, the ED component is important to maintain confidence that infection has not been re-introduced since the last survey was carried out.
As a general guideline, the hazard situation influences the emphasis placed on ad hoc surveys:
- Post-endemic: After elimination of an endemic disease or an outbreak, the likelihood of residual infection is still relatively high. Therefore, surveillance with the objective of FFD is important to be included.
- Free: If disease has never been present or has been absent for a long time, then the risk of residual infection becomes negligible so that activities focus more strongly on the risk of new introductions and thus early detection.
For hazards which can be easily detected via passive surveillance (e.g. FMD), demonstration of disease freedom in the “free” situation can purely rely on passive surveillance. However, legal requirements may still require that ad hoc surveys are carried out to provide additional confidence in freedom e.g. to satisfy demands by trading partners or stakeholders.
Early detection
- Relevant measure: a) Probability of detecting a significant increase (outbreak) of disease (outbreak detection), b) probability of detecting a disease if present at a certain level (prevalence) (presence)
- Coverage: Comprehensive coverage is essential to ensure outbreaks are not missed (usually achieved using passive surveillance), risk-based if active surveillance can increase the likelihood of detection
- Means of data acquisition: Active, passive or a combination of active and passive (depending on risk of introduction)
- Frequency of sampling: Continuous (e.g. passive surveillance), surveys at short intervals (e.g. monthly or weekly) or targeted surveys during the high risk period to ensure that infection is detected as quickly as possible; length of interval depends on disease impact, consequences, etc.
- Testing method: Detection of pathogen generally preferred for confirmation as it allows earlier detection of new introductions; however, detecting exposure by means of serological surveillance may be used instead or in combination as it is more sensitive once the “window period” has been passed
- Design prevalence: Very low as the aim is to detect introductions early (e.g. 0.01%); depends on prevalence at source and risk of introduction; a conservative estimate means choosing a design prevalence which tends towards zero to increase sample size and thus sensitivity
- Frequency of analysis: Continuous
Freedom from disease (FFD)
- Relevant measure: Probability of detecting an existing infection in case the population is infected at or above the specified design prevalence
- Coverage: Risk-based approaches are particularly suitable as focussing resources on high-risk population strata increases the likelihood of detection and thus the value of the information, which results in more cost-effective designs; generally combined with passive surveillance to provide comprehensive coverage as part of early detection
- Means of data acquisition: Active (often complemented by passive components aimed at early detection)
- Frequency of sampling: Repeated surveys
- Testing method: Detection of immune response preferred
- Design prevalence: Generally higher than for ED (e.g. 0.1%, 1%, 5%); in many cases legal requirements specify the design prevalence; a conservative estimate means choosing a design prevalence which tends towards zero
- Frequency of analysis: After each survey
Prevalence estimation (PREV)
- Relevant measure: Prevalence in an infected population
- Coverage: Structured surveys to obtain representative estimates. Risk-based approaches require exact information about risk ratios, which is rarely available, to take into account any resulting bias at the analytical stage
- Frequency of sampling: One-off (estimate prevalence) or repeated surveys (estimate changes in prevalence, e.g. to monitor the effect of intervention measures)
- Means of data acquisition: Active; passive surveillance can be used as complementary information, but it is often difficult to account for bias
- Testing method: Detection of immune response preferred as it is more practical and often less costly
- Design prevalence: According to the expected prevalence; a conservative estimate means choosing a design prevalence which tends towards 50% to maximize sample size and thus increase precision (e.g. if prevalence is estimated to range between 5% and 20%, then choose 20%)
- Frequency of analysis: After each survey
Case detection (CD)
- Relevant measure: Probability of detecting cases (existing and new infections) within an infected population
- Coverage: Often risk-based to increase the likelihood of detection; if the aim is to eliminate the disease then comprehensive coverage is required
- Frequency of sampling: Continuous sampling or periodic surveys (frequency could be risk-based, i.e. more frequent sampling of high risk herds)
- Means of data acquisition: Active (needed if elimination is the aim) or passive components
- Testing method: Detection of pathogen or immune response
- Design prevalence: Depends on the pathogen and duration of immunity; a conservative estimate means choosing a design prevalence which tends towards zero to increase sample size and thus sensitivity
- Frequency of analysis: Continuously
Note:
These recommendations on surveillance approaches based on the chosen objective are only general guidelines, which need to be applied whilst keeping the disease of interest, the design details and the circumstances in mind. Here are some exceptions to illustrate situations for which other approaches are adequate than those listed above.
Examples of exceptions:
Frequency of sampling:
- Continuous sampling at the abattoir may serve FFD, as the abattoir may just be an easily accessible sampling point (e.g. Trichinella);
- Due to practical and ethical criteria, hunting seasons exist so that sampling is restricted to those times of the year; despite this limitation, hunting may serve as active surveillance method for early detection.
Testing method:
- Also depends on whether the aim is to measure the percentage of exposed herds (measuring immune response) or the percentage of herds with ongoing infection (detection of the pathogen);
- For some disease the detection window for virology is very short (e.g. FMD, CSF) or delay in detection is expected to be long (e.g. surveillance of hunted wild boar), so that serology may be used instead or in combination with serological testing in case of ED;
- Other diseases result in lifelong immunity so that serology is not useful to assess the current disease situation in a previously exposed animal; however, serology may still be useful at the herd level to monitor new infections in seronegative replacement stock;
- For some disease-species combinations, serological testing is not possible (e.g. lack of commercially available ELISA tests for wild bird surveillance), so that pathogen detection methods may also applied to demonstrate disease freedom.
The terms ‘evaluation’ and ‘assessment’ are not true synonyms, although there could be an overlap and/or confusion in their use, both in the literature and among the various actors involved in evaluation. ‘Evaluation’ includes judgement of the system and/or recommendations to strengthen it, whereas ‘assessment’ is more a technical exercise, involving measurement of indicators.
An evaluation can occur at any development stage of a surveillance system: during the planning, design or implementation stages or ex-poste.
Ideally, evaluations should be carried out regularly, their frequency depending on the context and objective of the surveillance. However, one-off evaluations are possible in certain circumstances.
An evaluation can consider one, several or all components in the surveillance system. The extent of the evaluation depends on the evaluation question.
Examples:
The evaluation published by Sandberg et al. (2002) focussed on two of the various components of the Norwegian Salmonella surveillance and control programme: the systematic sampling in swine breeding herds and random sampling of pig carcasses at the abattoirs.
Any number of attributes can be considered by an evaluation, depending on the evaluation question. The number of attributes included has an impact on resources and complexity and should therefore be considered carefully.
Examples
- Evaluation of the surveillance of Bovine Enzootic Leucosis in Australian dairy cattle. One attribute considered: sensitivity (Hutchison and Martin 2005)
- Evaluation of the surveillance of West Nile Virus in British Columbia. One attribute considered: representativeness (David et al. 2007).
- Evaluation of a military syndromic surveillance system set up in French Guyana. Seven attributes considered: timeliness, data quality, acceptability, usefulness, stability, portability and flexibility (Jefferson et al. 2008).
- Economic evaluation of the surveillance and intervention programmes for bluetongue and BVD respectively (Hasler, Howe, Presi, et al. 2012; Hasler, Howe, Di Labio, et al. 2012)
A value judgement is part of the evaluation process but can be performed either by the evaluators or by the decision makers requesting the evaluation. It can be obtained by the confrontation of the system against targets set by the stakeholders, standard criteria or against a counterfactual. This step needs to be transparent and objective.
Examples.
- An evaluation of the scrapie surveillance in Great-Britain lead to the conclusion that the sensitivity of the system was poor and recommended that the possibility to implement other surveillance activities was investigated (Vidal Diez, Arnold, and Del Rio Vilas 2009).
- An evaluation published by Betanzos-Reyes et al. (2007) compared two alternative systems for the surveillance of malaria in Mexico, concluding that one had a higher cost-effectiveness than the other.
Evaluators can be internal and/or external, depending on the evaluation question.
Betanzos-Reyes, Angel Francisco, Mario Henry Rodríguez, Luis Gerardo Duran-Arenas, Juan Eugenio Hernández-Avila, Jorge Fernando Méndez-Galván, Oscar Julio Velázquez Monroy, and Roberto Tapia Coyner (2007). 'Comparative analysis of two alternative models for epidemiological surveillance in the Mexican Malaria Control Program', Health Policy, 80: 465-82.
Cameron, A. (2012). Manual of basic animal disease surveillance. Nairobi, Kenya, African Union: Interafrican Bureau for Animal Resources.
David, ST, S Mak, L MacDougall, and M Fyfe (2007). 'A bird's eye view: using geographic analysis to evaluate the representativeness of corvid indicators for West Nile virus surveillance', International Journal of Health Geographics, 6: 3.
Dorea, F. C., J. Sanchez and C. W. Revie (2011). "Veterinary syndromic surveillance: Current initiatives and potential for development." Preventive Veterinary Medicine 101(1-2): 1-17.
Dufour, B. and G. Hendrikx (2009). Epidemiological surveillance in animal health, Food and Agriculture Organization.
Dupuy, C., A. Bronner, E. Watson, L. Wuyckhuise-Sjouke, M. Reist, A. Fouillet, D. Calavas, P. Hendrikx and J. B. Perrin (2013). "Inventory of veterinary syndromic surveillance initiatives in Europe (Triple-S project): Current situation and perspectives." Preventive Veterinary Medicine 111(3-4): 220-229.
Enhanced Passive Surveillance project, The FAZD Center.
Garcia-Abreu, A., W. Halperin and I. Danel (2002). Public health surveillance toolkit: A guide for busy task managers, World Bank.
Hadorn, D. C. and K. D. C. Stärk (2008). "Evaluation and optimization of surveillance systems for rare and emerging infectious diseases." Veterinary research 39(6): 1-12.
Häsler, B., et al. (2011). "Conceptualising the technical relationship of animal disease surveillance to intervention and mitigation as a basis for economic analysis." Bmc Health Services Research 11.
Häsler, B., K. S. Howe, E. Di Labio, H. Schwermer, and K. D. Stark (2012). 'Economic evaluation of the surveillance and intervention programme for bluetongue virus serotype 8 in Switzerland', Prev Vet Med, 103: 93-111.
Häsler, B., K. S. Howe, P. Presi, and K. D. Stark (2012). 'An economic model to evaluate the mitigation programme for bovine viral diarrhoea in Switzerland', Prev Vet Med, 106: 162-73.
Hoinville, L. J., L. Alban, J. A. Drewe, J. C. Gibbens, L. Gustafson, B. Haesler, C. Saegerman, M. Salman and K. D. C. Staerk (2013). "Proposed terms and concepts for describing and evaluating animal-health surveillance systems." Preventive Veterinary Medicine 112(1-2): 1-12.
Hoinville, L. J., et al (2013). Animal Health Surveillance Terminology.
Hutchison, Jenny, and Tony Martin (2005). "An evaluation of surveillance for enzootic bovine leucosis in the Australian dairy industry". In.: Australian Government, Department of Agriculture, Fisheries and Forestry.
Jefferson, H., B. Dupuy, H. Chaudet, G. Texier, A. Green, G. Barnish, J. P. Boutin, and J. B. Meynard (2008). 'Evaluation of a syndromic surveillance for the early detection of outbreaks among military personnel in a tropical country', J Public Health (Oxf), 30: 375-83.
OIE (2010). Handbook on import risk analysis for animals and animal products. Volume 1: Introduction and qualitative risk analysis. Paris, France, OIE.
OIE (2014a). Guide to terrestrial animal health surveillance. Paris, France, OIE.
OIE (2014b). "Terrestrial animal health code." Retrieved 19 March 2013, online.
Rodriguez-Pietro, V., et al. (2014). "Systematic review of surveillance systems and methods for early detection of exotic, new and re-emerging diseases in animal populations." Epidemiology and Infection: 1-25.
Sandberg, Marianne, Petter Hopp, Jorun Jarp, and Eystein Skjerve (2002). 'An evaluation of the Norwegian Salmonella surveillance and control program in live pig and pork', International Journal of Food Microbiology, 72: 1-11.
Schatz, J., C. M. Freuling, E. Auer, H. Goharriz, C. Harbusch, N. Johnson, I. Kaipf, T. C. Mettenleiter, K. Muhldorder, R. U. Muhle, B. Ohlendorf, B. Pott-Dorfer, J. Pruger, H. S. Ali, D. Stiefel, J. Teubner, R. G. Ulrich, G. Wibbelt and T. Muller (2014). "Enhanced Passive Bat Rabies Surveillance in Indigenous Bat Species from Germany - A Retrospective Study." Plos Neglected Tropical Diseases 8(5).
The TripleS Project: Syndromic Surveillance Systems in Europe.
Vial, F. and M. Reist (2014). "Evaluation of Swiss slaughterhouse data for integration in a syndromic surveillance system." Bmc Veterinary Research 10.
Vidal Diez, A., M.E. Arnold, and V.J. Del Rio Vilas (2009). "Evaluation and optimization of scrapie surveillance in Great Britain: a Bayesian framework." In Proceedings of the 12th Meeting of the International Society of Veterinary Epidemiology and Economics, 10-14 August 2009. Durban, South Africa.
WHO (2005). "WHO Recommended Surveillance Standards." WHO/CDS/CSR/ISR/99/2/EN.
